
For Sharvil Patel, leading Zydus Lifesciences has meant strengthening the company’s scientific base while expanding its global presence across healthcare segments.
Patel has focused on building what he describes as a strong innovation engine across businesses with sustained investment in research. “We have consistently invested 7–8% revenues on innovation over the years,” says Patel.
Investments support research programmes across new chemical entities (NCEs), biotechnology, vaccines, rare disease therapies, generics and wellness products. “All our businesses are supported by a robust innovation engine and a network of regulatory-compliant manufacturing facilities across the globe,” says Patel.
Under his leadership, Zydus Lifesciences operates across pharmaceuticals, consumer wellness and med-tech, serving patients in more than 90 countries. “We are an innovation-led global life-sciences company with an aim to transform lives by offering diverse healthcare solutions through the entire patient journey and empowering people with the freedom to live healthier and more fulfilled lives,” says Patel.
In NCE research, referring to medicines discovered and developed through a company’s own scientific work, the company became the first Indian pharmaceutical firm to develop and launch its own molecule, Saroglitazar.
“We have so far launched two NCEs in India from our own pipeline,” says Patel. Saroglitazar has progressed into global development. The molecule has completed Phase II(b)/III trials for a primary biliary cholangitis indication in the United States. Commercial preparation is under way.
Another therapy, Desidustat, has been licensed for China, where marketing approval is awaited.
For Patel, the winner of the Services Vanguard category in pharmaceuticals and healthcare sector in the BT-PWC India’s Best CEOs list, programmes like these illustrate how sustained research capability can support development of medicines from discovery to their launch in global markets.

Focus on biologics, vaccines
Alongside chemical research, Patel has expanded the company’s work in biotechnology and vaccines. “On the biotech front, we have one of the largest biosimilar portfolios in India with 16 biosimilars approved and 13 launches in India,” he says.
The company has also secured marketing authorisations for biosimilars in multiple emerging markets. “We have 10 biosimilars under development including seven oncology biosimilars,” says Patel.
Vaccines is another area of focus. “On the vaccines front, we have marketing authorisation for 17 vaccines; eight are under development. Two of our vaccines are WHO pre-qualified,” says Patel.
While innovation programmes attract attention, Patel often highlights the generics business as the backbone of the company’s operations. “The US is our largest market,” he says.
Over the years, the company has consistently filed between 25 and 30 abbreviated new drug applications (ANDAs) annually. “So far, we have made over 480 ANDA filings and received over 420 approvals,” says Patel. The development pipeline currently includes around 200 products.
This foundation also allows the company to continue investing in innovation programmes while maintaining financial stability.

Analysts tracking the company say the next phase of growth will depend on how the pipeline translates into launches. In a recent report, Param Desai and Kushal Shah of Prabhudas Lilladher said the company “is working on a robust pipeline of complex products, including injectables, 505(b)(2), transdermals, NCEs, biosimilars and vaccines,” though these opportunities are “expected to materialise over the next two–three years.” They add the company is “looking for three–four sizeable low competition opportunities in US,” noting that “timely launch will be the key to sustaining momentum in US sales.”
Alongside internal research, Patel has also relied on partnerships and acquisitions to strengthen the company’s development pipeline. One such move was the acquisition of LiqMeds in 2023. The company focuses on liquid oral formulations developed through the 505(b)(2) regulatory pathway.
These formulations are particularly useful for paediatric and geriatric patients who may have difficulty swallowing tablets. The company has also in-licensed biosimilar candidates such as pembrolizumab and ranibizumab as part of its expansion in global markets.
Beyond prescription medicines, Patel has also laid strong emphasis on expanding the company’s consumer wellness business. “In our existing business, we hold leadership position across multiple product categories in India,” he says.
The segment operates with more than 2,600 field personnel and nearly 2,000 distributors covering more than 2.6 million retail outlets across the country.

More recently, the company entered the digital vitamins and supplements market through the acquisition of the US-based firm Comfort Click. “Comfort Click has a strong presence in the UK and the EU and is building its presence in the US,” says Patel.
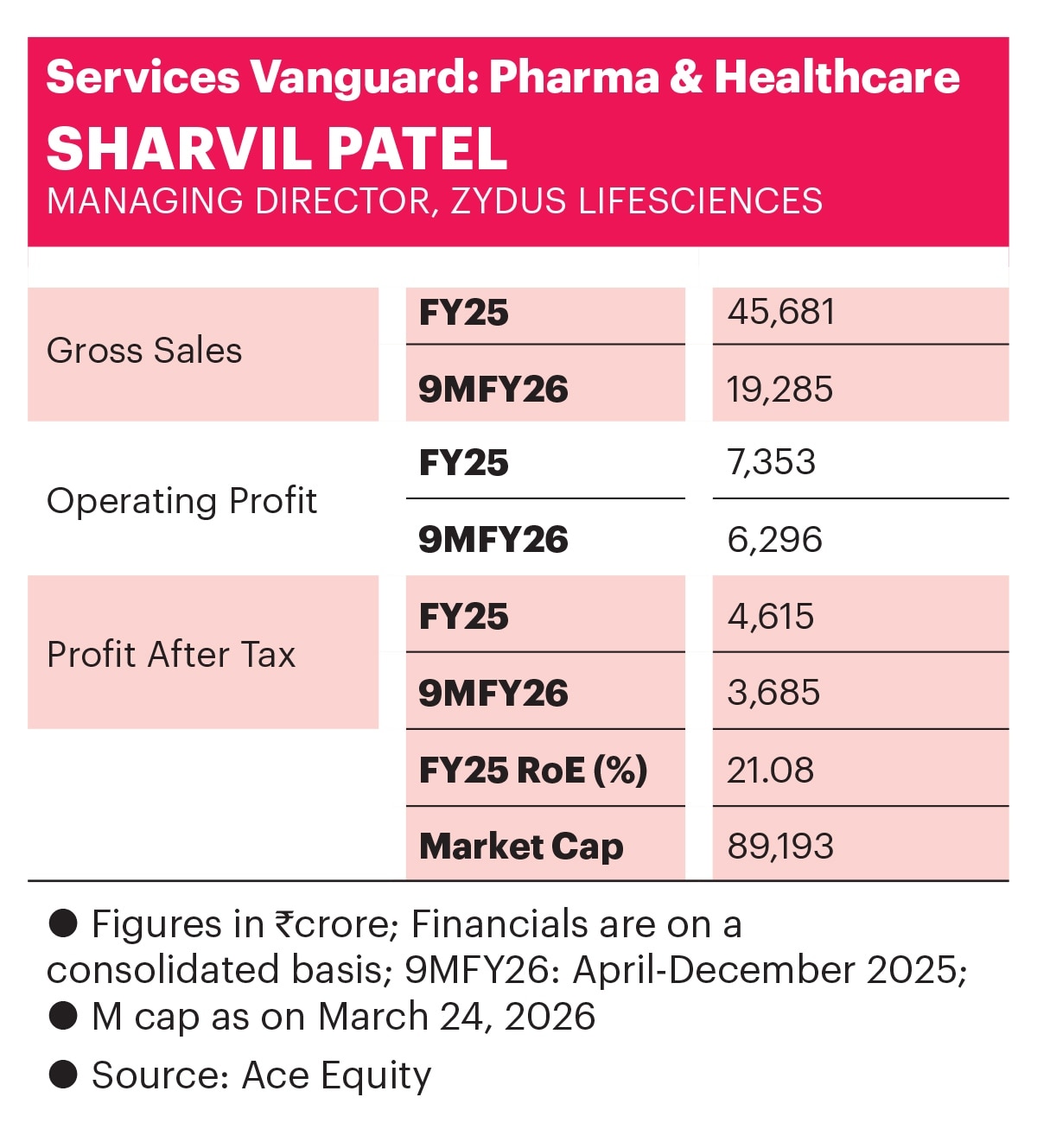
The company’s financial performance over the past three years reflects the outcome of these strategic efforts. In FY25, profit after tax was Rs 4,615 crore, on gross sales of Rs 45,681 crore. Return on equity was a healthy 21%.
Patel highlights the company’s workforce profile. Around 61% employees are under 35.
He says this plays a role in strengthening research and innovation capabilities, digital tools, and data-driven approaches.
In the future, advancing innovation pipelines in regulated markets and strengthening the presence across global healthcare segments will continue to guide the company’s direction.
@neetu_csharma