
The World Health Organization (WHO) said on Wednesday the only approved vaccine against malaria should be widely given to African children, potentially marking a major advance against a disease that kills hundreds of thousands of people annually.
The WHO recommendation is for RTS,S - or Mosquirix - a vaccine developed by British drugmaker GlaxoSmithKline (GSK.L).
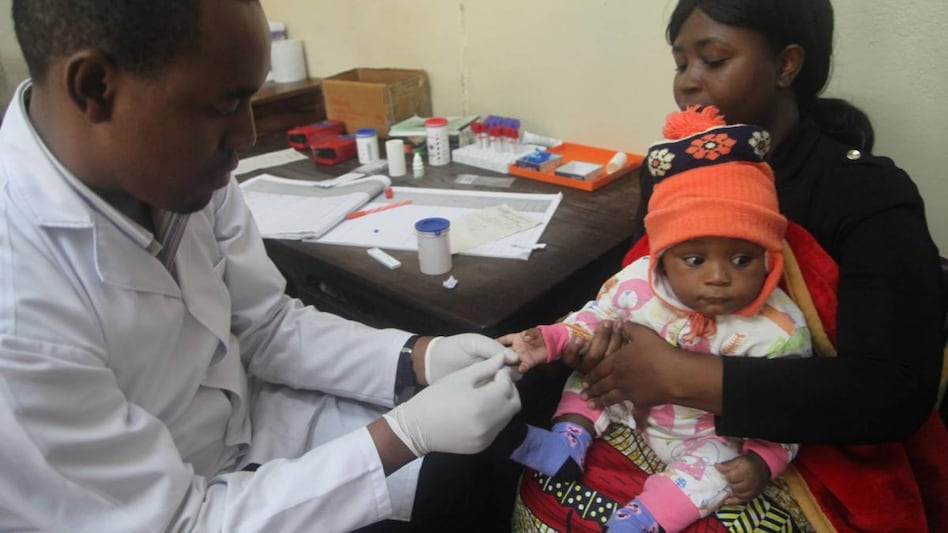
Since 2019, 2.3 million doses of Mosquirix have been administered to infants in Ghana, Kenya and Malawi in a large-scale pilot programme coordinated by the WHO. The majority of those whom the disease kills are under age five.
That programme followed a decade of clinical trials in seven African countries.
"This is a vaccine developed in Africa by African scientists and we're very proud," said WHO director-general Tedros Adhanom Ghebreyesus.
"Using this vaccine in addition to existing tools to prevent malaria could save tens of thousands of young lives each year," he added, referring to anti-malaria measures like bed nets and spraying to kill mosquitoes that transmit the disease.
One of the ingredients in the Mosquirix vaccine is sourced from a rare evergreen native to Chile called a Quillay tree. Reuters reported on Wednesday that the long-term supply of these trees is in question.
Malaria is far more deadly than COVID-19 in Africa. It killed 386,000 Africans in 2019, according to a WHO estimate, compared with 212,000 confirmed COVID-19 deaths in the past 18 months.
The WHO says 94% of malaria cases and deaths occur in Africa, a continent of 1.3 billion people. The preventable disease is caused by parasites transmitted to people by the bites of infected mosquitoes. Symptoms include fever, vomiting and fatigue.
The vaccine's effectiveness at preventing severe cases of malaria in children is only around 30%, but it is the only approved vaccine. The European Union's drugs regulator approved it in 2015, saying its benefits outweighed the risks.
"This is how we fight malaria, layering imperfect tools on top of each other," said Ashley Birkett, who leads global malaria vaccine work at Path, a non-profit global health organization that has funded development of the vaccine with GSK and the three-country pilot.
Another vaccine against malaria called R21/Matrix-Mthat was developed by scientists at Britain's University of Oxford showed up to 77% efficacy in a year-long study involving 450 children in Burkina Faso, researchers said in April. It is still in the trial stages.
GSK welcomed the WHO recommendation.
"This long-awaited landmark decision can reinvigorate the fight against malaria in the region at a time when progress on malaria control has stalled," Thomas Breuer, GSK's chief global health officer, said in a statement.
GSK shares held steady in New York following the announcement, which came after the close of trading in its London-listed shares.