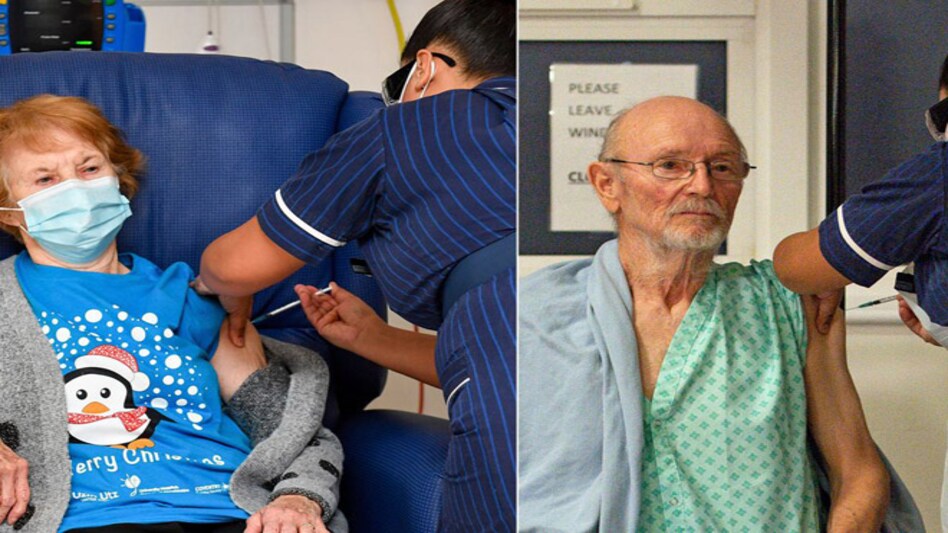
 Margaret Keenan, 90, (left) and William Shakespeare, 81, were the first and second recipients of Pfizer's COVID-19 vaccine
Margaret Keenan, 90, (left) and William Shakespeare, 81, were the first and second recipients of Pfizer's COVID-19 vaccine Margaret Keenan, 90, (left) and William Shakespeare, 81, were the first and second recipients of Pfizer's COVID-19 vaccine
Margaret Keenan, 90, (left) and William Shakespeare, 81, were the first and second recipients of Pfizer's COVID-19 vaccineBritish healthcare regulator has issued a warning that people with history of significant allergic reactions should not be given the newly approved Pfizer-BioNTech COVID-19 vaccine. The advisory was issued after two NHS workers suffered allergic reactions on Tuesday.
Medicines and Healthcare products Regulatory Agency (MHRA) said that the allergy advisory applies to people who have had reactions to medicines, food or vaccines, reported the BBC.
ALSO READ: Pfizer vaccine: UK regulator allays fears after 4 Bell's palsy cases in US trials
Dr June Raine, chief executive of MHRA, informed a parliamentary committee on Wednesday that the cases of severe allergic reactions did not appear during vaccine trials. "We know from the very extensive clinical trials that this wasn't a feature. But if we need to strengthen our advice, now that we've had this experience in the vulnerable populations - the groups have been selected as a priority - we get that advice to the field immediately," said Dr Raine.
The two UK healthcare workers reportedly developed anaphylactoid reaction shortly after the Pfizer COVID-19 vaccine was administered to them, the report said. They received treatment shortly after, and are stable now.
Anaphylactoid reactios appear as skin rashes, breathlessnes and a drop in blood pressure. This is different from anaphylaxis, which can lead to death.
ALSO READ: William Shakespeare second person to get Pfizer's COVID-19 vaccine
Notably, both individuals have prior history of allergies and carried adernaline pens for fiirst aid in the event of severe reactions.
The Pfizer-BioNTech jab, codenamed BNT162b2, became the first ever vacccine against coronavirus to receive emergency use approval - first in the UK and then in Bahrain. The companies have also approved regulators in India for emergency use authorisation of the inoculation, but it seems like a long shot, considering the fact it has never underwent any trials in the country. The vaccine has to be stored at extremely frigid temperatures of -70 degree Celsius, which presents another argument against its appoval by the Indian regulator.
Meanwhile, the UK has began to vaccinate its priority groups against COVID-19 using the Pfizer vaccine. Margaret Keenan, 90, and William Shakespeare, 81, were the first and second recipients of the jab vaccine in the world. The British government intends to administer 800,000 in the coming weeks.
ALSO READ: COVID-19 vaccine: 3 out of 24,000 participants react adversely to Oxford-AstraZeneca candidate