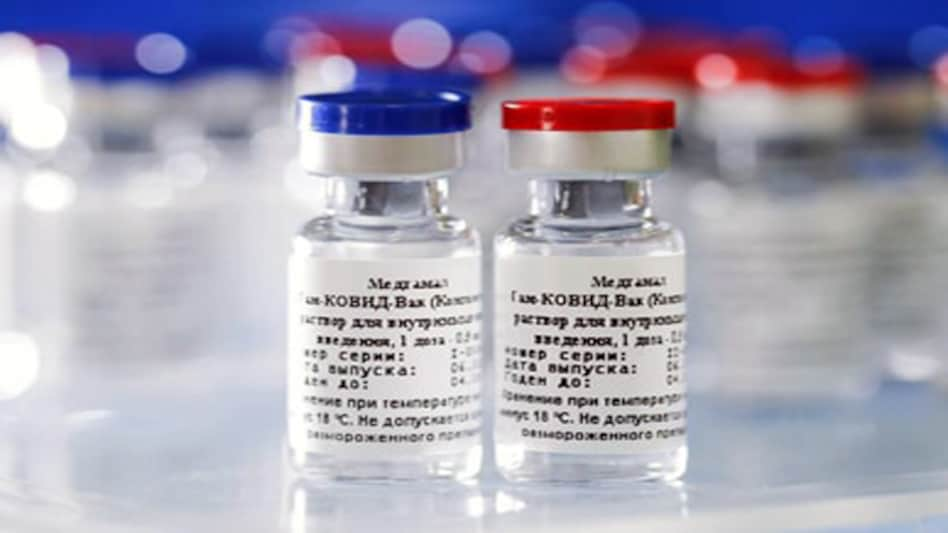

Dr Reddy's Laboratories announced on Tuesday it has commenced Phase 2 and 3 clinical trials for Sputnik V vaccine in association with Russian Direct Investment Fund (RDIF) in India.
"Dr. Reddy's Laboratories Ltd and Russian Direct Investment Fund (RDIF) announced today that they have commenced adaptive phase 2/3 clinical trials for Sputnik V vaccine in India after receiving the necessary clearance from the Central Drugs Laboratory, Kasauli, India. This will be a multicenter and randomized controlled study, which will include safety and immunogenicity study," Dr Reddy's said in a stock exchange filing.
The trials are being conducted by JSS Medical Research, being the clinical research partner. Dr. Reddy's has also joined hands with Biotechnology Industry Research Assistance Council (BIRAC), Department of Biotechnology (DBT) for advisory support, as well as to use BIRAC's clinical trial centres for the vaccine.
Also Read: TCS deploys supply chain solution to double India's COVID-19 testing capacity
Recently, RDIF announced the second interim results of its vaccine candidate's clinical trial data, which showed 91.4% efficacy on day 28 after the first dose; and efficacy over 95% on 42 days after the first dose. As of now, there are 40,000 volunteers partaking in Phase 3 trials of the virus, of which over 22,000 have already been vaccinated with the first dose, and over 19,000 have received both doses.
G.V. Prasad, Co-chairman and Managing Director at Dr. Reddy's said, "This is another significant step as we continue to collaborate with multiple entities along with the government bodies to fast-track the process for launching the vaccine in India. We are working towards making the vaccine available with a combination of import and indigenous production model." Dr. Reddy's and RDIF had partnered up in September 2020, so as to conduct clinical trials of the Sputnik V vaccine, as well as having the rights for distribution of its first 100 million doses in India.
The Sputnik V vaccine developed by the Gamaleya National Research Institute of Epidemiology and Microbiology was registered by the Ministry of Health of Russia on August 11 this year making it the world's first registered vaccine (based on the human adenoviral vector platform) against the pandemic.
Also Read: Unilever experiments four-day working week in New Zealand