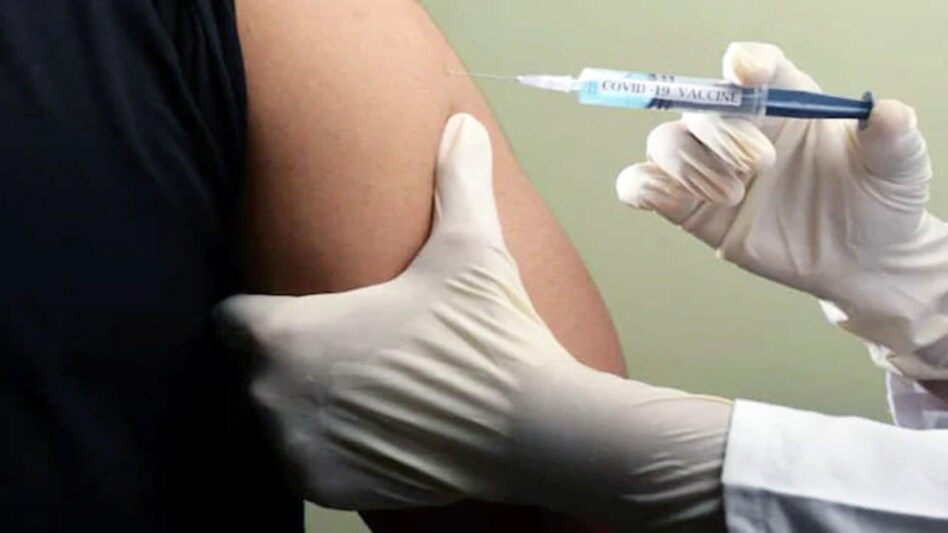
 Vaccine manufacturers have been permitted to release up to 50 per cent of their supply to state governments and in the open market.
Vaccine manufacturers have been permitted to release up to 50 per cent of their supply to state governments and in the open market. Vaccine manufacturers have been permitted to release up to 50 per cent of their supply to state governments and in the open market.
Vaccine manufacturers have been permitted to release up to 50 per cent of their supply to state governments and in the open market.In the view of the rising COVID-19 cases in the country, the government on Monday announced that from May 1 everyone above the age of 18 years would be eligile to get the vaccination against the virus.
Vaccine manufacturers have been permitted to release up to 50 per cent of their supply to state governments and in the open market at a pre-declared price.
States have also been empowered to procure additional vaccine doses directly from the manufacturers, as well as open up vaccination to any category of people above the age of 18 years for the same.
Also read: Former PM Manmohan Singh tests COVID-19 positive, rushed to AIIMS
The decision to start the Phase-III of vaccination programme was taken during a meeting chaired by Prime Minister Narendra Modi. "In its Phase-III, the National Vaccine Strategy aims at liberalised vaccine pricing and scaling up of vaccine coverage. This would augment vaccine production as well as availability, incentivising vaccine manufacturers to rapidly ramp up their production as well as attract new vaccine manufacturers, domestic and international," the government said in a release.
While vaccine manufacturers would have to supply 50 per cent of their monthly Central Drugs Laboratory (CDL) released doses to the Government of India, they would be free to supply the remaining 50 per cent doses to state governments and in the open market.
"Manufacturers would transparently make an advance declaration of the price for 50 per cent supply that would be available to state governments and in open market, before 1st May 2021. Based on this price, state governments, private hospitals, industrial establishments etc would be able to procure vaccine doses from the manufacturers. Private hospitals would have to procure their supplies of Covid-19 vaccine exclusively from the 50 per cent supply earmarked for other than Government of India channel," it said.
Private vaccination providers will have to transparently declare their self-set vaccination price. The eligibility through this channel would be opened up to all adults, i.e. everyone above the age of 18, the government said.
Also read: COVID-19: Gennova Biopharma to start mRNA vaccine trials this month
The Centre's vaccination programme will continue as before, providing free of cost vaccination to health care workers (HCWs), front line workers (FLWs) and all people above 45 years of age.
"All vaccination (through Government of India and other than Government of India channel) would be part of the National Vaccination Programme, and mandated to follow all protocol such as being captured on CoWIN platform, linked to AEFI (adverse events following immunisation) reporting and all other prescribed norms. Stocks and price per vaccination applicable in all vaccination centres will also have to be reported real-time," the government said.
While 50-50 per cent distribution of vaccine to the Centre and others will be applicable uniformly across for all vaccines manufactured in the country, imported vaccines will be allowed to be utilised in the other than Government of India channel.
The Centre will allot vaccines from its shares to states and union territories based on the criteria of extent of infection and performance based on speed of administration. "Wastage of vaccine will also be considered in this criteria and will affect the criteria negatively. Based on the above criteria, state-wise quota would be decided and communicated to the states adequately in advance," it said.
"Second dose of all existing priority groups i.e. HCWs, FLWs and population above 45 years, wherever it has become due, would be given priority, for which a specific and focused strategy would be communicated to all stakeholders," the release added.
India is reeling under the second wave of coronavirus cases, with scarcity of hospital beds, ventilators, oxygen, medicines being reported from across the country. The country has administered 12.38 crore of COVID-19 vaccine doses as of Monday morning.
In January, the Centre had given emergency use approval to Serum Institue of India's Covishield vaccine and Bharat Biotech's Covaxin to fight against the COVID-19 pandemic. Earlier this month, Russia's Sputnik V was also given approval, while the Centre also fast-tracked approval process for foreign made COVID-19 vaccines which have been approved by drug regulators in US, UK, Europe and Japan.
Also read: NASA's 'Wright Brothers moment'; flies first helicopter on Mars