 The Drugs Controller General of India (DCGI) has approved teh emergency use of drug 2-deoxy-D-glucose (2-DG) developed by DRDO
The Drugs Controller General of India (DCGI) has approved teh emergency use of drug 2-deoxy-D-glucose (2-DG) developed by DRDO The Drugs Controller General of India (DCGI) has approved teh emergency use of drug 2-deoxy-D-glucose (2-DG) developed by DRDO
The Drugs Controller General of India (DCGI) has approved teh emergency use of drug 2-deoxy-D-glucose (2-DG) developed by DRDOThe Drugs Controller General of India (DCGI) on Saturday gave approval to the anti-COVID drug developed by DRDO (Defence Research and Development Organisation) for emergency use.
The drug, 2-deoxy-D-glucose (2-DG), to treat coronavirus patients has been developed by the Institute of Nuclear Medicine and Allied Sciences (INMAS), a lab of Defence Research and Development Organisation (DRDO), in collaboration with Dr Reddy's Laboratories, Hyderabad.
"Clinical trial results have shown that this molecule helps in faster recovery of hospitalized patients and reduces supplemental oxygen dependence. Being a generic molecule and analogue of glucose, it can be easily produced and made available in plenty. Higher proportion of patients treated with 2-DG showed RT-PCR negative conversion in Covid patients," DRDO said in an official statement on Saturday.
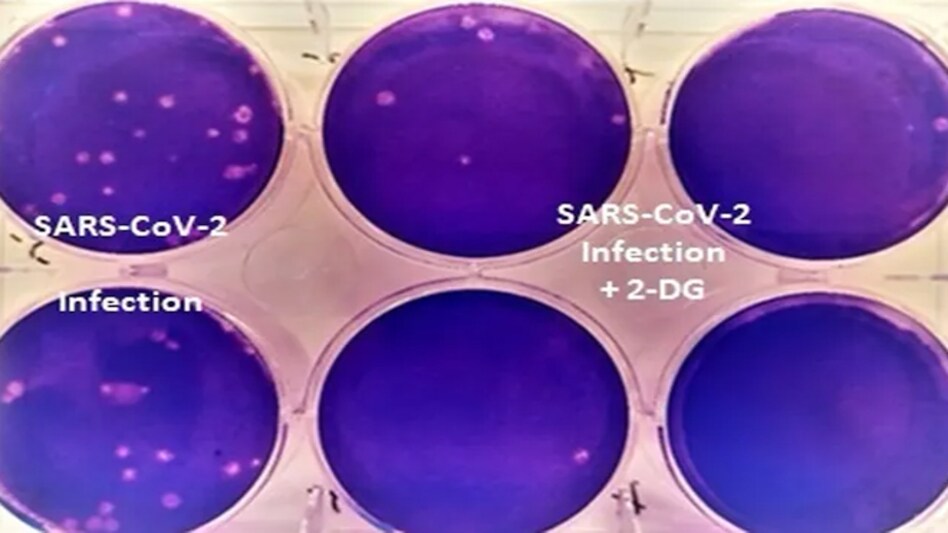
INMAS-DRDO scientists had in April 2020, during the COVID-19 first wave, carried out laboratory experiments with the Centre's help for Cellular and Molecular Biology (CCMB), Hyderabad. The researchers found that this molecule works effectively against the SARS-CoV-2 virus and impedes viral growth.
Also Read: COVID-19 crisis: IAF, DRDO come to the rescue as India faces acute oxygen shortage
On the basis of these findings, DCGI's Central Drugs Standard Control Organization (CDSCO) granted approval for phase-II clinical trial of 2-DG in COVID-19 patients in May 2020. The drug was found to be safe in COVID-19 patients in phase-II trials conducted during May to October 2020. It also manifested considerable improvement in the patients' recovery.
"In efficacy trends, the patients treated with 2-DG showed faster symptomatic cure than Standard of Care (SoC) on various endpoints. The drug comes in powder form in sachet, which is taken orally by dissolving it in water. It accumulates in the virus-infected cells and prevents virus growth by stopping viral synthesis and energy production," the statement added.
On the basis of successful results, DCGI further allowed the phase-III trials in November 2020.