 (Picture Courtesy: Youtube @PIB India)
(Picture Courtesy: Youtube @PIB India) (Picture Courtesy: Youtube @PIB India)
(Picture Courtesy: Youtube @PIB India)Indian health regulators are looking at four more COVID-19 vaccines, said Health Secretary Rajesh Bhushan on Tuesday.
Among these vaccines are those developed by Zydus Cadila, Biological E and Gennova, along with Russia's Sputnik-V vaccine.Zydus Cadila finished Phase 2 clinical trials in December last year and has been granted approval for Phase 3 trials in India.
Similarly, the Phase 2 clinical trials of Russia's Sputnik-V COVID-19 vaccine have also concluded and Phase 3 trials are being carried out by its Indian partner Dr Reddy's Laboratories.
Biological E started Phase 1 clinical trials of its vaccine in December and Phase 2 is expected to kick-off in March. Gennova's RNA-based COVID-19 vaccine is currently in Phase 1 with Phase 2 clinical trials likely to begin in March of this year.
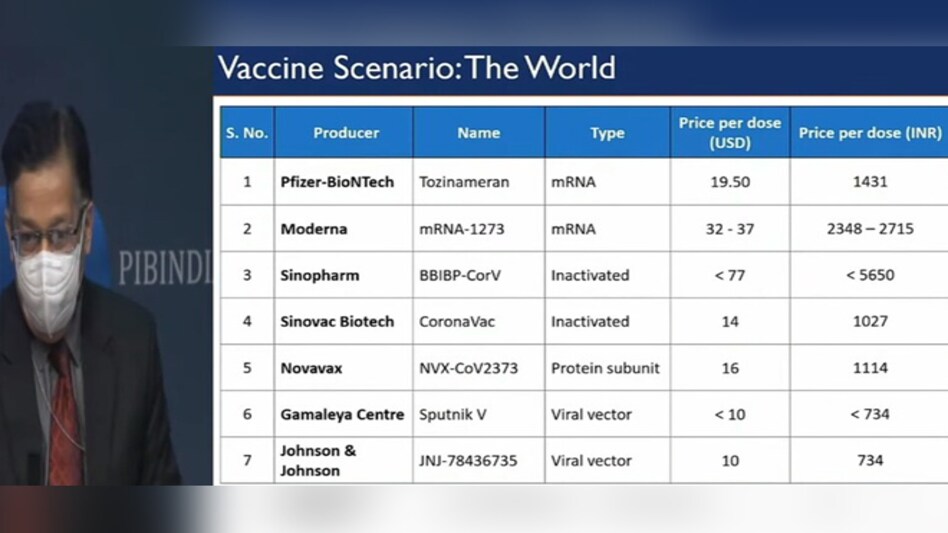
Sputnik-V, the COVID-19 vaccine developed by Russia's Gamaleya Research Institute of Epidemiology and Microbiology, will cost Rs 734 per dose, same as developed by Johnson & Johnson is also expected to be priced at Rs 734 per dose.
Pfizer-BioNTech's COVID-19 vaccine will be priced at Rs 2,800 for two doses while Moderna's will cost around Rs 2,300 to Rs 2,700 per dose.
One vaccine developed by China has been priced at Rs 5,600 per dose while another Chinese vaccine will be made available in India at Rs 1,200 per dose.The Oxford-AstraZeneca's vaccine manufactured by Serum institute of India (SII) will cost the government Rs 200 a vial.
The Health Secretary added that Hyderabad-based Bharat Biotech's coronavirus vaccine Covaxin is being purchased at a cost of Rs 206 per dose by the Government of India.
The COVID-19 situation is very worrisome across the world, Health Secretary Rajesh Bhushan said during a press briefing, adding that active cases continue to decline in India. "Now, active cases are 2,16,558. This sort of number was last seen on June 30, 2020," he said.
Asked about the vaccines approved by Indian regulators, Health Secretary Rajesh Bhushan said both Covishield and Covaxin have established safety and immunogenicity. "All preparations are on track for vaccine rollout. It will be a sequential rollout, depending on availability," he added.
The government has approved two vaccines for emergency use so far - Oxford-AstraZeneca's Covishield and Bharat Biotech's Covaxin.
Also read: 5.7 crore Covishield doses to be shipped today; here's how much these 13 cities will get
Also read: 'Covishield, Covaxin both part of vaccination drive; 5,000 vaccine points,' says Dr Vinod Paul